1. Radicular origins and neuro-functional role
The pudendal nerve arises from the anterior branches of the sacral roots S2, S3, and S4. This pluriradicular origin is a key feature: it combines somatic fibers (sensory and motor) with fibers from the autonomic nervous system, particularly parasympathetic fibers (Robert et al., 1998).
This dual component explains why pudendal neuralgia is not limited to localized pain. Nerve irritation may manifest as burning sensations or electric shocks, but also as urinary, anorectal, or sexual functional disturbances, without any visible lesion of the involved organ (Labat et al., 2008/2023).
The S2–S4 roots contribute to storage and evacuation functions (bladder and rectum) as well as perineal sensitivity. Any mechanical constraint applied to the pudendal nerve may therefore produce disproportionate sensory and functional consequences (Labat et al., 2008/2023).
2. A sinuous pathway with high mechanical vulnerability
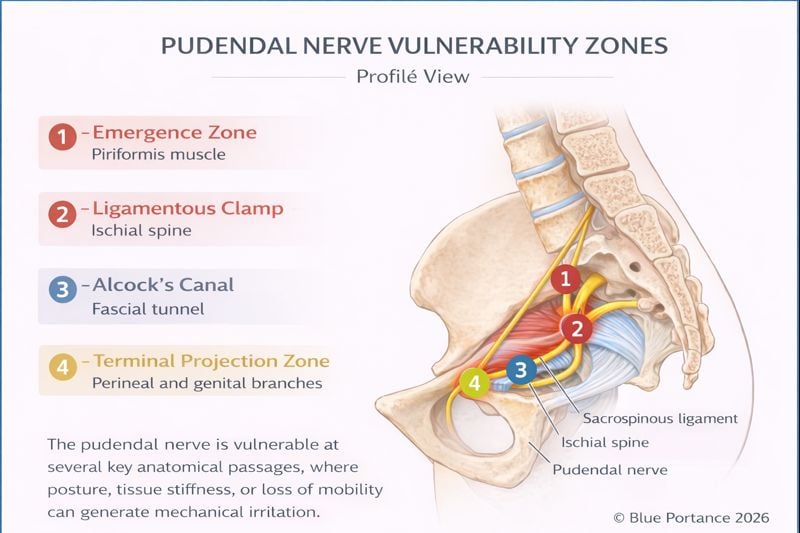
The pudendal nerve follows a distinctive course: it exits the pelvic cavity only to re-enter it almost immediately. This anatomical detour, essential to its functional role, also represents a biomechanical vulnerability (Robert et al., 1998).
After its emergence, the nerve leaves the pelvis through the greater sciatic notch, curves around the ischial spine, and then re-enters the pelvis through the lesser sciatic notch to travel within Alcock’s canal (Robert et al., 1998).
This pathway forces the nerve to pass through several narrow anatomical zones, sensitive to postural variations, muscle tension, and pelvic geometry. With each change in position—particularly during prolonged sitting—the nerve may be subjected to variable shear and compression forces (Labat et al., 2008/2023).
3. Terminal branches and referred pain logic
At the end of its main course, the pudendal nerve divides into three terminal branches, commonly referred to as the pudendal trident (Labat et al., 2008/2023).
- Inferior rectal nerve: innervates the anal canal and may explain anorectal pain, foreign body sensations, or deep burning.
- Perineal nerve: innervates the urethra and perineum; it may be associated with urinary or perineal burning without identifiable infection (Labat et al., 2008/2023).
- Dorsal nerve of the clitoris or penis: provides fine genital sensation; it may be associated with electric shocks, hypersensitivity, or contact-related pain.
This organization explains why pain is often perceived “at a distance” from the actual conflict: the brain interprets the nociceptive signal at the terminal zone, while the mechanical constraint may be located higher along the nerve pathway (Labat et al., 2008/2023).
4. The nerve within its fascial environment
The pudendal nerve does not “float” freely within the pelvis: it is intimately connected to the pelvic fasciae, particularly the obturator internus fascia, which contributes to the formation of Alcock’s canal (Cholewa et al., 2024).
Fasciae allow physiological neural gliding. When the fascial matrix densifies, tissue mobility decreases: the nerve may be mechanically stressed during each micro-movement, even in the absence of visible anatomical lesions (Stecco & Stecco, 2024).
Normal imaging does not exclude nerve suffering: part of the problem may relate to impaired gliding and an altered tissue environment (Stecco & Stecco, 2024).
5. Adaptive reserve and decompensation threshold
The pudendal nerve has a certain adaptive capacity: it can tolerate transient constraints without pain. Neuralgia develops when the cumulative burden (posture, mechanical constraints, muscle tension, fascial densification) exceeds an individual adaptive reserve (Labat et al., 2008/2023).
This concept is essential to understanding why some individuals develop neuralgia without identifiable trauma, and why pain may progressively settle over time.
- Labat, J.-J., Riant, T., Robert, R., et al. (2008; updated 2023). Diagnostic criteria for pudendal neuralgia by pudendal nerve entrapment (Nantes criteria).
- Robert, R., Prat-Pradal, D., Labat, J.-J., et al. (1998). Anatomical basis of pudendal nerve entrapment. Surgical and Radiologic Anatomy.
- Cholewa, J., et al. (2024). Myofascial entrapment in Alcock’s canal: the role of pelvic floor hypertonia.
- Stecco, A., & Stecco, C. (2024). Fascial densification and neuropathic pain: clinical implications.


